One of the joys about the learning to run experiments process is that one gets to learn the results of both intended and unintended phenomena. In my experiments the intention is to seal powder of known composition into gold capsules along with a sufficient H2O and graphite to ensure that the chemical reactions which take place when we elevate the pressure and temperature (to simulate what happens to rocks buried at great depth) take place in “water-saturated” conditions (which is to say there is enough water available for the growth of minerals which require water as part of their chemical formula, such as the micas). However, learning to weld the capsules is a difficult process (I’ve got a draft post on that topic just waiting for me to get photos that actually display the features I want them to show). As a result of my welding trials and tribulations I’ve had mixed success in the “sealing” part of the above paragraph. Despite the issues with my first attempts at sealing, we ran my first experiment nonetheless, giving two samples a week and a half at elevated pressure/temperature (in this case 650 C and 25 kbars). Once they were “cooked” we had the gold capsules mounted into small disks of epoxy, then carefully polished the disks until the insides of the capsules were exposed. During the polishing stage we received our first confirmation that they had not achieved the same level of “sealed”. Apparently when properly sealed the presence of water inside the capsules ensures that the pore space in between the grains of powder are occupied, and as a result even the high pressures to which we subject them aren’t enough for the new minerals to properly interlock when they grow. As a result, while there are new crystals present, the texture isn’t very rock-like, and when polishing it is easy to accidentally remove clumps of the sample itself. This is the texture we were anticipating, and, for one of the samples run in the first experiment, this is exactly what happened. In these cases we polish only enough to just expose the inside of the capsule, then add more epoxy, letting it soak down into those pore spaces and let it dry before completing the polishing process without so much risk in losing what we are trying to polish.
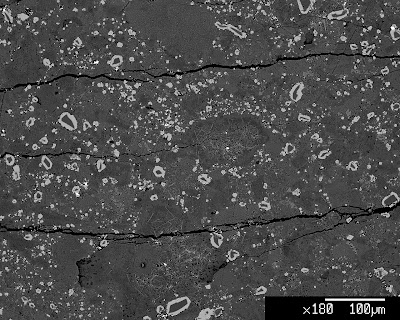
However, in the other of the two samples run in the first experiment I must not have done the final welding properly, because the contents of the capsule were much harder, and held together better, meaning that the pore space was not held open with fluid when the minerals were growing. This was obvious during the polishing process, so I was able to go quite a bit deeper into the capsule (remember these are only 2 mm in diameter and about 5 mm long so “deeper” is only a relative term) before needing to add the additional epoxy.
Today we got to look at these samples in the microprobe, and as expected from the difference in their textures noted while polishing them, they are rather different from one another. The one wherein I had issues with the welding did contain some water; we know this because there are very small grains of mica present. However, neither was it water-saturated, so it lost some due to the poor seal of the capsule. It contains many, many very tiny grains of garnet (~1 micron diameter; remember that there are 1000 microns in every millimeter) which nucleated on their own, and very thin rims of garnet on the “seeds” which had been included in the powder to encourage garnet growth. The rest of the sample is even finer grained “matrix” minerals, which are going to be difficult to analyze. The other, water saturated, sample contains fewer, larger, grains of garnet, and the rims of new garnet growth on the “seeds” are much thicker than in the first sample. While it, too, is generally fine-grained, it will be easier to find single crystals large enough to get a good analysis of their compositions (which we need if we are going to accomplish our goals).
Having had this first look at the samples we’ve set the probe to create “element maps”, pretty full-colour pictures showing which areas are high (warm colours) and which areas are low (cool colours) in specific elements. Once we have these maps, we will use them to select the grains for the detailed compositional analysis. But even before we do that, I now have a better understanding of the difference between water-saturated and water-under saturated environments in terms of the ease at which minerals grow.